 
MDU matrix and their quantitative determinations. 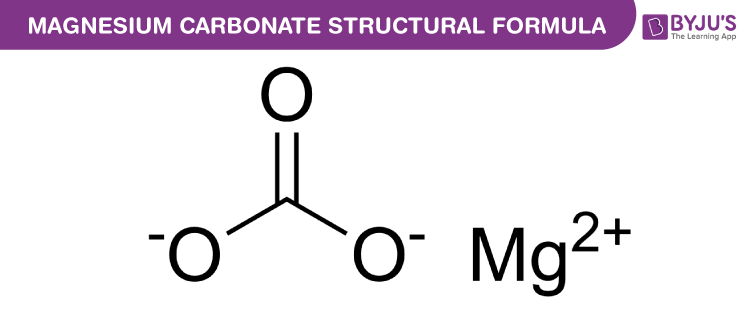
In inert and air environments for the impurity characterization of Thermal analysis with simultaneous TG–DTA–EGA measurements 
Literature was found scarce, if carbon and carbonates both are present as impurities in the matrix and required The quantitative determination of carbon and carbonates in the Though combustion method is in use for the determination of carbon present in the matrix, it cannot be applied for Transformation Infrared Spectrometric system for the analysis ofĮvolved gases. Simultaneous TG–DTA measurements become a powerful tool if coupled with mass spectrometric or Fourier The properties most often studied are specific heatĪnd enthalpy changes, mass loss or mass gain, thermal expansionĪnd gas evolution. Physico-chemical changes by application of programmed temperature variation. Thermoanalytical methods are useful to systematically analyze the Stoichiometric composition but the concentration of the compound should be between 2% and 5% in the matrix. Technique can be used to identify the impurity with respect to its To provide any information regarding the stoichiometric composition of the impurity present in the matrix. Moreover, the limitation of these techniques lie in their inability Na and Mg, which may cause interference in the measurements. However, these techniques cannot be applied directly to yellowĬake samples due to the presence of other major elements like Interference from the matrices like uranium and its oxides. Matrix separation is essential before the determination of analytes due to the problem of either chemical or spectral Spectrometric techniques are being used for the trace element Specifications, quality assurance of MDU is essential. Levels of impurities are being encountered in the leach solution In the processing of low-grade ores, higher In yellow cake to examine whether their levels are confirming to V, Mo, B, Cd, REEs, Th, chloride, sulphate and phosphate is required Determination of U, Mg, Na, K, Fe, Mn, Si, Al, Cd, Cr, Cu, Co, Uranium ore using dilute sulphuric acid and subsequent ion-exchange separation of trace impurities from the leach solution followed by magnesium diuranate precipitation using magnesia Magnesium diuranate (yellow cake) is the intermediate product Most frequently encountered are the uranates (M2UO4) and diuranates (M2U2O7). Oxides with alkali, alkaline earth and a number of other metal ions. Uranium in its hexavalent state forms ternary Magnesium oxide, sodium hydroxide and hydrogen peroxide haveīeen effective. A number of different precipitants such as ammonia, PH range, acid or alkaline, depending on the solution type and precipitant used. In general, uranium in theįorm of yellow cake can be precipitated from solution over a wide Uranium as nuclear fuel has stringent specifications regarding Also these studies confirm the formation of magnesium uranate, uranium sesquioxide and uranium dioxide above 1000 ☌, due to the decomposition of magnesium diuranate. Powder X-ray diffraction analysis of magnesium diuranate suggests the presence of magnesium hydroxide as impurity in the Quantitatively using TG and FTIR techniques and the results are in good agreement. CarbonĪnd magnesium hydroxide content present as impurities in magnesium diuranate have been determined Through the evolved gas analysis using Fourier Transformation Infrared Spectrometry detection. Presence of carbonate and sulphate as impurities in the matrix was confirmed The stoichiometric composition of MDU was identified as MgU2O7#H2O. With respect to the impurities present in the matrix. – evolved gas analysis techniques for the compositional characterization of magnesium diuranate (MDU) Reddy aĪnalytical Chemistry Division, B.A.R.C., Mumbai 400 085, IndiaĬurrent studies describe the application of simultaneous thermogravimetry–differential thermal analysis Journal homepage: Impurity characterization of magnesium diuranate using simultaneous Journal of Nuclear Materials 436 (2013) 40–46Ĭontents lists available at SciVerse ScienceDirect 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
